 |
 |
|
|
|
|
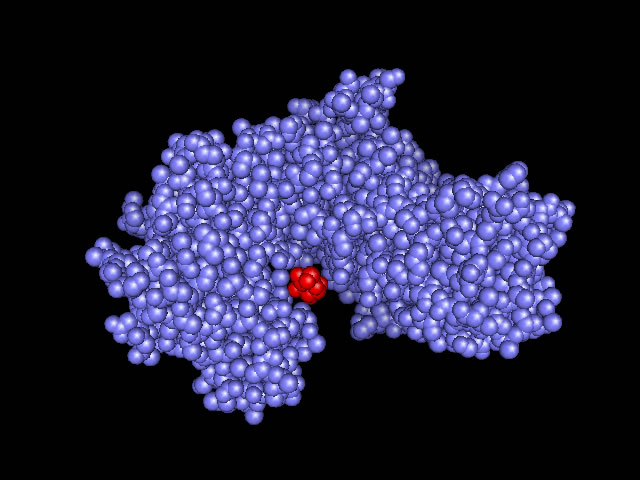
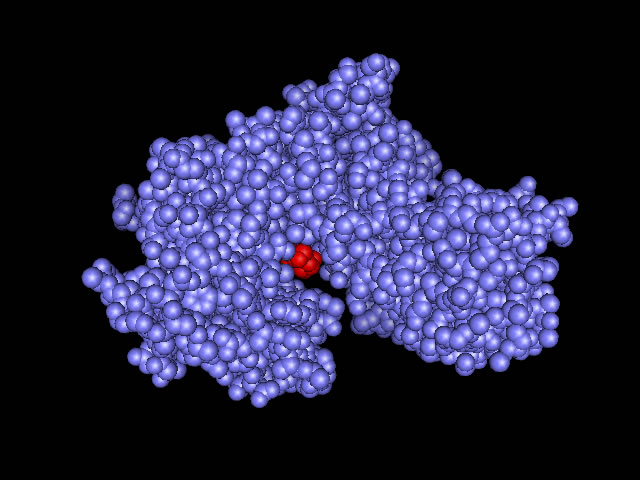
Lehningers definition of induced fit: A change in the conformation of an enzyme in response to substrate binding that renders the enzyme catalytically active. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| This is the home page of Daniel E. Koshland http://mcb.berkeley.edu/faculty/BMB/koshlandd.html |
| (requires CHIME to observe computer graphics) This site shows the glucose binding sites of hexokinase, hydrophobic channels, and the open and closed forms of hexokinase in three dimensional diagrams. http://www.siu.edu/departments/biochem/chime_rasmol/glycolysis/hexokinase_comp.htm |
| (requires CHIME to observe computer graphics) This site has an interactive animation of the mechanism and conformational changes of our enzyme before and after binding of its substrate (glucose). http://www.wiley.com/legacy/college/boyer/0470003790/structure/hexokinase/hexokinase.htm |
| This site shows a tutorial on the induced fit model with an emphasis on the mechanisms kinetics. http://bmbiris.bmb.uga.edu/wampler/8010/lectures/kinetics/steady/sld073.htm |
| This site contains not only a virtual tutorial of induced fit, but also contains links to additional bodies of information on the subject. http://resources.ed.gov.hk/biology/english/health/enzyme/enzyme_factors_inducedfit.html |
| This site illustrates a simple 2D animation of induced fit. It also includes how a substrate will approach and then bind to the active site of an enzyme. http://bio.winona.msus.edu/berg/ANIMTNS/ind-fit.htm |
| (requires CHIME to observe computer graphics) This site shows an interactive model of the enzyme hexokinase, and then Wireframe model of hexokinase. http://scholar.hw.ac.uk/site/biology/activity7.asp?outline |
| Here is a link to a power point slide show which contains information and images on the following topics for human brain hexokinase: biological significances, enzyme function, enzyme structure, binding site, binding mechanism, and enzyme kinetics. http://www.chem.uncc.edu/courses/6101/Lectures/Student%20Presentations/ATPBINDINGSITEOFHUMANBRAINHEXOKINASE.ppt |
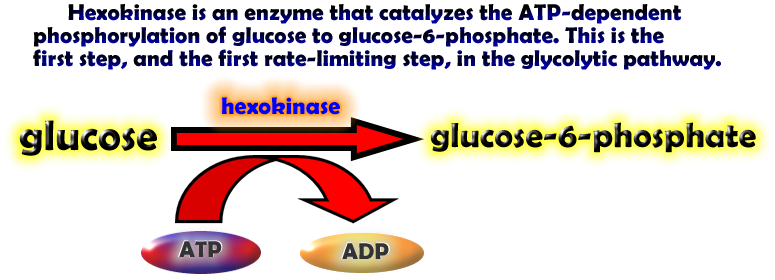
| The Protein Data Banks Molecule of the Month picture of hexokinase and short summery explaining its significance in the first step of the glycolysis. http://www.rcsb.org/pdb/molecules/pdb50_2.html |
| This is a link to The Protein Families Database of Alignments under the topic of hexokinase. http://www.sanger.ac.uk/cgi-bin/Pfam/getacc?PF00349 |
| Glucose-Induced Conformational Change in Yeast Hexokinase W S Bennett, Jr and T A Steitz; Proceedings of the National Academy of Sciences of the United States of America, 1978, vol. 75: 48484852 http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=336218 |
| The Key-Lock Theory and the Induced Fit Theory Daniel E. Koshland, Jr.; Chemistry International Education England, 1994, vol. 33: 2375-2378 http://www.tulane.edu/~biochem/lecture/gbch701/koshland2.pdf |
| Conformational Changes of Proteins Arising From Normal Mode Calculations F. Tama and Y.-H. Sanejouand; Protein Engineering, 2001, vol. 14: 1-6 http://www.tulane.edu/~biochem/lecture/gbch701/koshland2.pdf |
| Induced Fit in Guanidino kinasesComparison of Substrate-Free and Transition State Analog Structures of Arginine Kinase Mohammad S. Yousef, Shawn A. Clark, Pamela K. Pruett, Thayumanasamy Somasundaram, W. Ross Ellington, and Michael S. Chapman; Protein Science, 2003, vol. 12: 103-111 http://www.proteinscience.org/cgi/content/full/12/1/103 |
| Proteomics and Models for Enzyme Cooperativity Daniel E. Koshland Jr. and Kambiz Hamadani; Journal of Biological Chemistry, 2002, vol. 277: 46841-46844 http://www.jbc.org/cgi/content/full/277/49/46841 |
| Structural Model of Human Glucokinase in Complex With Glucose and ATP Bhuvaneshwari Mahalingam, Antonio Cuesta-Munoz, Elizabeth A. Davis, Franz M. Matschinsky, Robert W. Harrison, and Irene T. Weber; Diabetes, 1999, vol. 48: 1698-1705 http://diabetes.diabetesjournals.org/cgi/reprint/48/9/1698 |